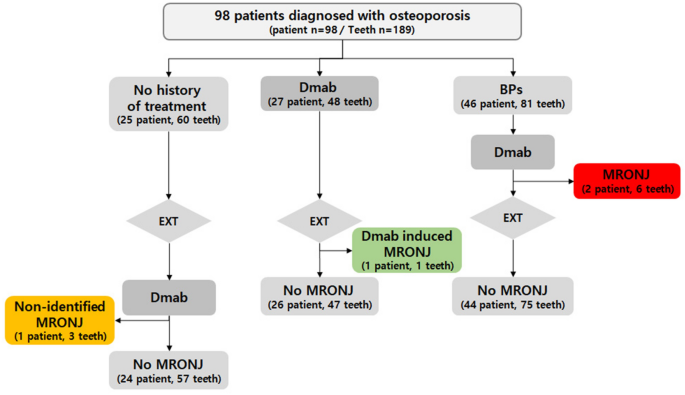
Combination denosumab and high dose teriparatide for postmenopausal osteoporosis (DATA-HD): a randomised, controlled phase 4 trial - The Lancet Diabetes & Endocrinology

Combination denosumab and high dose teriparatide for postmenopausal osteoporosis (DATA-HD): a randomised, controlled phase 4 trial - The Lancet Diabetes & Endocrinology
Denosumab 60mg injection (Prolia®) for Osteoporosis in Post- menopausal Women and Men (over 50 years) in Primary Care Prescribi